In, atomic packing factor (APF), packing efficiency or packing fraction is the fraction of volume in a that is occupied by constituent particles. It is dimensionless and always less than unity. In systems, by convention, the APF is determined by assuming that atoms are rigid spheres. The radius of the spheres is taken to be the maximal value such that the atoms do not overlap.
For one-component crystals (those that contain only one type of particle), the packing fraction is represented mathematically bywhere N particle is the number of particles in the unit cell, V particle is the volume of each particle, and V unit cell is the volume occupied by the unit cell. It can be proven mathematically that for one-component structures, the most dense arrangement of atoms has an APF of about 0.74 (see ), obtained by the. For multiple-component structures, the APF can exceed 0.74. Single component crystal structuresCommon taken on by atomic systems are listed below with their corresponding packing fraction. (hcp): 0.74.
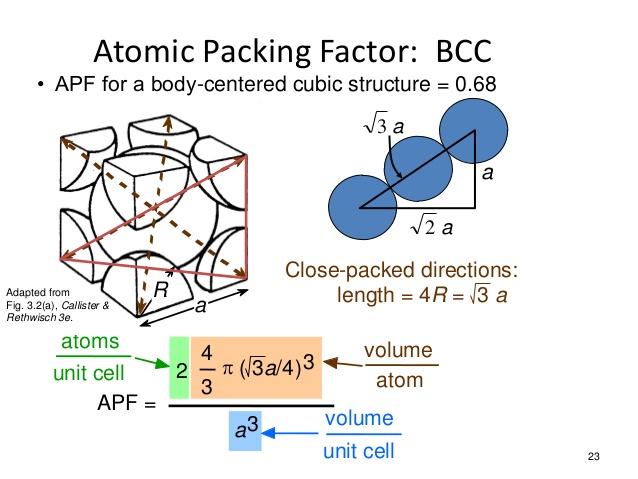
3.5 We are asked to show that the atomic packing factor for BCC is 0.68. The atomic packing factor is defined as the ratio of sphere volume to the total unit cell volume, or APF = VS VC Since there are two spheres associated with each unit cell for BCC VS = 2(sphere volume) = 2 4πR3 3 ⎛ ⎝ ⎜ ⎜ ⎞ ⎠ ⎟ ⎟ = 8πR3 3. Simple Cubic, fcc and bcc. There are three cubic structures that general chemistry students are taught. They are called simple cubic, face-centred cubic, and body-centred cubic. They vary in how the atoms/spheres are arranged inside of it. Simple Cubic.
(fcc): 0.74 (also called cubic close-packed, ccp). (bcc): 0.68.: 0.52.: 0.34The majority of metals take on either the hcp, ccp or bcc structure. Body-centered cubic. BCC structureThe for the crystal structure contains several fractions taken from nine atoms: one on each corner of the cube and one atom in the center. Because the volume of each of the eight corner atoms is shared between eight adjacent cells, each BCC cell contains the equivalent volume of two atoms (one central and one on the corner).Each corner atom touches the center atom.
A line that is drawn from one corner of the cube through the center and to the other corner passes through 4 r, where r is the radius of an atom. By geometry, the length of the diagonal is a √ 3. Therefore, the length of each side of the BCC structure can be related to the radius of the atom byKnowing this and the formula for the, it becomes possible to calculate the APF as follows: Hexagonal close-packed. Ellis, Arthur B.; et al.
Teaching General Chemistry: A Materials Science Companion (3rd ed.). Washington, DC: American Chemical Society.
084122725X. Moore, Lesley E.; Smart, Elaine A. Solid State Chemistry: An Introduction (3rd ed.). Boca Raton, FL: Taylor & Francis, CRC. 8.Further reading. Schaffer; Saxena; Antolovich; Sanders; Warner (1999). The Science and Design of Engineering Materials (2nd ed.).
New York, NY: WCB/McGraw-Hill. 81–88. Callister, W.
Materials Science and Engineering (6th ed.). San Francisco, CA: John Wiley and Sons.
DescriptionIn crystallography, atomic packing factor ( APF), packing efficiency or packing fraction is the fraction of volume in a crystal structure that is occupied by constituent particles. It is dimensionless and always less than unity.
In atomic systems, by convention, the APF is determined by assuming that atoms are rigid spheres. The radius of the spheres is taken to be the maximal value such that the atoms do not overlap. For one-component crystals (those that contain only one type of particle), the packing fraction is related to the number of particles in the unit cell, the volume of each particle, and the volume occupied by the unit cell.
( For one-component structures, the most dense arrangement of atoms has an APF of about 0.74).Hexagonal close-packed (hcp): 0.74Face-centered cubic (fcc): 0.74 (also called cubic close-packed (ccp))Body-centered cubic (bcc): 0.68Simple cubic: 0.52Diamond cubic: 0.34.
